|
Alternate Anode Reaction for Copper Electrowinning
The conventional copper electrowinning process uses the water hydrolysis reaction as the anodic source of electrons However this reaction generates acid and oxygen gas The oxygen which leaves the system is associated with some acid from the electrolyte resulting in an acid mist |
|
ARSENIC’S BEHAVIOUR AND BENEFITS IN COPPER ELECTROREFINING
The concentration of arsenic in anodes varies widely depending on the copper source The typical anode arsenic concentration reported by 35 copper refineries is presented in Figure 1 (Moats Robinson Wang Filzwieser Siegmund & Davenport 2013) Anode composition ranges from 7 to 1800 ppm with an average of 870 |
What is electrolytic refining?
In this setup, an electrolyte (metal salt aqueous solution), depending on the metal, is often used. The clean or pure metal is formed at the cathode when the electrical current of a sufficient voltage is applied by dissolving impure metal at the anode. Electrolytic refining is also sometimes referred to as electrorefining.
Can low-grade chalcopyrite be used for copper production?
A vision is proposed in which an electrolytic technique, integrated with low cost and sustainable power, enables the production of unprecedented annual tonnages of copper from low-grade chalcopyrite, with valuable by-products and enhanced selectivity for impurities. 1. Introduction
Can copper be purified by electrolytic refining?
Even small concentrations of impurities noticeably lower copper’s electrical conductivity. Such a high degree of purity can be obtained by electrolytic refining in a cell similar to that shown in Figure 17.5.1 17.5. 1. Figure 17.5.1 17.5. 1: The electrolytic purification of copper.
How can a high degree of purity be obtained by electrolytic refining?
Such a high degree of purity can be obtained by electrolytic refining in a cell similar to that shown in Figure 17.5.1 17.5. 1. Figure 17.5.1 17.5. 1: The electrolytic purification of copper. In such a cell a thin sheet of high-purity Cu serves as the cathode, and the anode is the impure Cu which is to be refined.
|
Presentación de PowerPoint
Recovery of copper by electrowinning. C. de la Parra1 M. Reig1 |
|
Presentación de PowerPoint
6 เม.ย. 2560 100 years of copper electrorefining this is an important stage in engineering design to make all stakeholders aware of hazards and many of ... |
|
Electrorefining Process of the Non-Commercial Copper Anodes
26 ก.ค. 2564 As the chemical dissolution of this form is more intense than the electrochemical one the copper concentration in the electrolyte increases |
|
The Life Cycle of Copper its Co-Products and By-Products
24 ม.ค. 2545 Electrowinning (EW): As noted above pure copper sulfate dissolved in sulfuric acid is generated by the vat leaching process and the solvent ... |
|
MINOR ELEMENTS IN COPPER SMELTING AND
The behavior of minor elements in electrorefining was described from a theoretica.1 viewpoint. In the matte smelting furnaces the volatilization of As |
|
National Pollutant Inventory (NPI) Emission Estimation Techniques
The process of electrowining copper differs from electro-refining as the copper deposits Ag Au ppt. Slag to Cu smelter. Anode casting. Ag crystals. Au mud. |
|
PowerPoint 프레젠테이션
23 พ.ย. 2565 1) Equivalent to expanding copper smelting capacity by up to 20000 tonnes (recycled copper production ... Electro Refining. Blister. (Cu 98% or ... |
|
Zero Emission Copper Mine of the Future
2 ก.ค. 2563 via electrorefining. Copper concentrates comprise the largest form ... South Australia Copper to the World Conference: Presentation. 80 BHP ... |
|
Electrodeposition and Electroless Deposition of Copper in
62 63 In copper electrowinning |
|
Metal Recovery by Electro Winning - A Product Concept
18 ก.พ. 2562 Copper electrowinning: theoretical and practical design. TWP. Matomo Process Plant South Africa |
|
Electrorefining of Copper
Electrorefining of Copper. 1. Electrorefining – General Introduction. In an electrorefining process the anode is the impure metal and the impurities must |
|
Copper Leach / Solvent Extraction / Electrowinning Technology
Key Facts About Copper Solvent Extraction Transfer copper from leach to electrowinning ... Variable Copper Concentration in Pregnant Leach Solution. |
|
Copper electrowinning: theoretical and practical design
For direct or post solvent extraction copper electrowinning design key theoretical considerations include current density and efficiency |
|
Copper Production Technology
for producing copper from exploration |
|
The World Copper Factbook 2021
The output is the same as through the electro-refining route - refined copper cathodes. ICSG estimates that in 2020 refined copper production from. SX-EW |
|
Electrometallurgy_Overview_Final_v2 _2_
Keywords: Electrometallurgy Electrowinning |
|
Hydrometallurgical processing of anode slime for recovery of
mum copper recovery in sulphuric acid and hydrochloric acid leaching electrorefining tank at the time of electrorefining of copper. It contains valuable. |
|
Optimizing the operating conditions of gold elution and
ELECTROWINNING FOR TAU LEKOA STREAM AT KOPANANG GOLD characterized by the presence of base metals like nickel copper |
|
Molten Salt Electrodeposition of Silicon in Cu-Si
This study proposed electrowinning of Si in molten Cu-Si alloy to decrease the grade silicon (SeG–Si) |
|
Cropsscutting Technology Development at the Center for Advanced
Azar 24 1384 AP the smelting |
|
Electrorefining of Copper - ct - ufrgs
Electrorefining of Copper 1 Electrorefining – General Introduction In an electrorefining process, the anode is the impure metal and the impurities must be |
|
Electrorefining of Copper
8 déc 1991 · level is 0 17 m-2d-1 were replaced with electrorefined material, with the result shown in Fig 212pb line at 238 4 keV The copper detector cap |
|
Electrodeposition and Electroless Deposition of Copper in
Here, the process is akin to copper electrowinning processes albeit with modified concentration was established, with the limit of detection in ppt level A pre-‐ |
|
Copper Leach / Solvent Extraction / Electrowinning Technology
Puts copper in an acid water solution • Solvent Extraction • Transfer copper from leach to electrowinning • Transfer acid from electrowinning to leach |
|
1 CHAPTER 1 INTRODUCTION This Section briefly describes the
chloride ions have been dosed in copper electrorefining for more than 100 years Figure 3-17: Schematic Presentation of the Concentration Profile of Cupric |
|
Copper electrowinning - SAIMM
For direct or post solvent extraction copper electrowinning design, key theoretical considerations include current density and efficiency, electrolyte ion |
|
Electrometallurgy_Overview_Final_v2 _2
Keywords: Electrometallurgy, Electrowinning, Electrorefining, Aluminum, Copper, Lead, Magnesium, Nickel, Titanium, and Zinc Abstract Electrolytic processing |
|
[PDF] Electrorefining of Copper - ct - ufrgs
Electrorefining of Copper 1 Electrorefining – General Introduction In an electrorefining process, the anode is the impure metal and the impurities must be |
|
[PDF] Electrorefining of Copper
Electrorefining of Copper R G H Robertson SNO STR 91 071 Dec 8, 1991 Abstract The electrorefining of copper to reduce the levels of natural alpha and |
|
[PDF] Electrometallurgy - MIT
ucts, whereas in an electrorefining cell the dissolution of the anode metal itself occurs The more noble metals such as copper and zinc are electrolyzed from |
|
[PDF] Copper electrowinning - SAIMM
For direct or post solvent extraction copper electrowinning design, key theoretical considerations include current density and efficiency, electrolyte ion |
|
[PDF] Copper Leach / Solvent Extraction / Electrowinning Technology
Puts copper in an acid water solution • Solvent Extraction • Transfer copper from leach to electrowinning • Transfer acid from electrowinning to leach |
|
[PDF] Electrodeposition and Electroless Deposition of Copper in
Here, the process is akin to copper electrowinning processes albeit with modified concentration was established, with the limit of detection in ppt level A pre ‐ |
|
[PDF] PDF 1MB
Keywords Electrometallurgy, Electrowinning, Electrorefining, Aluminum, Copper, Lead, Magnesium, Nickel, Titanium, and Zinc Abstract Electrolytic processing |
|
[PDF] 1 CHAPTER 1 INTRODUCTION This Section briefly describes the
Copper electrometallurgy includes electrorefining and electrowinning depending on whether the copper mineral was processed through pyrometallurgy or |
- Electrorefining of copper reaction
- Electrorefining of copper Class 10
- Electrorefining of copper diagram
- Electrorefining of gold
- Electrorefining of copper anode mud
- Explain the process of electrorefining of copper
- Electrolytic refining of copper
- Electrorefining of silver

PDF) Static-Bed Electrorefining of Copper Scrap Turnings
Source:https://d3i71xaburhd42.cloudfront.net/71f3600499b5b5a8e558aeb807129b6ffa9be9e2/12-Figure8-1.png
![PDF] Copper electrowinning: theoretical and practical design PDF] Copper electrowinning: theoretical and practical design](https://i1.rgstatic.net/publication/290474809_Examination_of_copper_electrowinning_smoothing_agents_Part_I_A_review/links/56d5175e08ae2cd682b94613/largepreview.png)
PDF] Copper electrowinning: theoretical and practical design
Source:https://i1.rgstatic.net/publication/290474809_Examination_of_copper_electrowinning_smoothing_agents_Part_I_A_review/links/56d5175e08ae2cd682b94613/largepreview.png

PDF) Examination of copper electrowinning smoothing agents Part I
Source:https://ars.els-cdn.com/content/image/1-s2.0-S001346860700802X-gr1.jpg
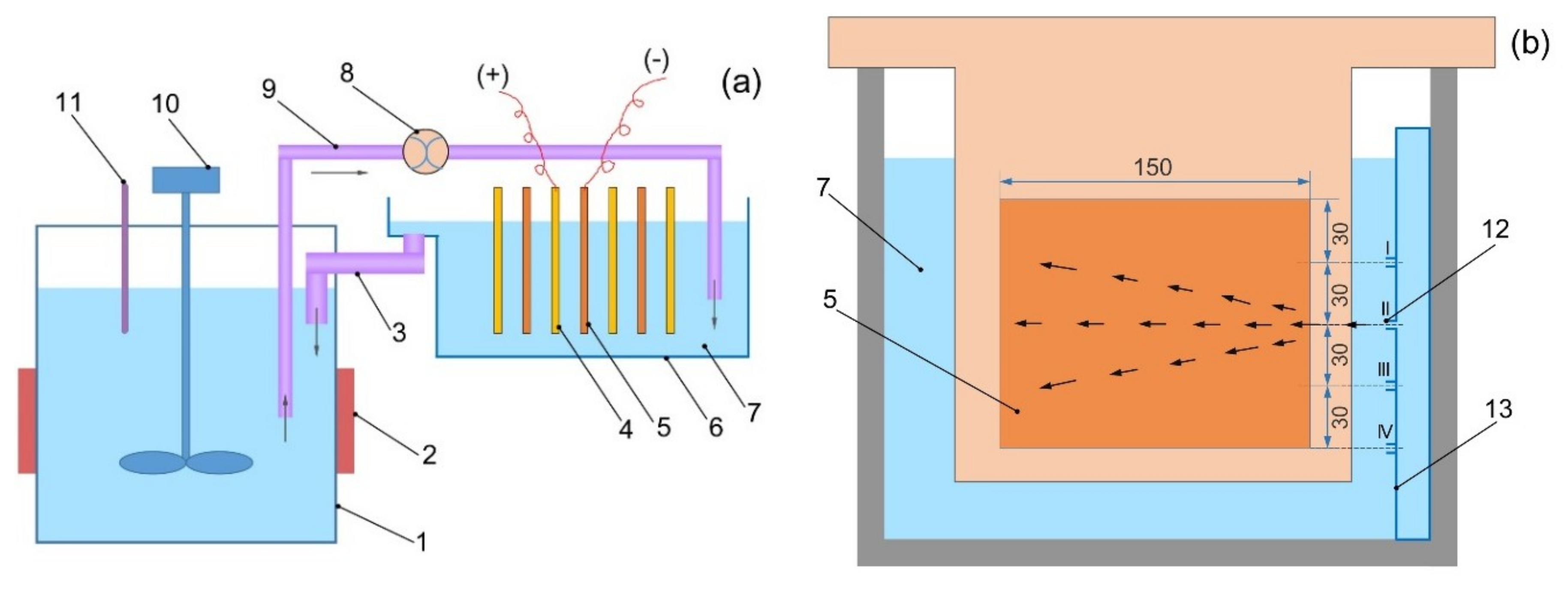
Influence of ammonium salt on electrowinning of copper from
Source:https://www.mdpi.com/metals/metals-08-00833/article_deploy/html/images/metals-08-00833-g001.png
Metals
Source: Free Full-Text

PDF) Electrowinning of copper from sulfate electrolyte in presence
Source:https://upload.wikimedia.org/wikipedia/commons/thumb/6/68/CuElectrolyticRefineApparatus.png/300px-CuElectrolyticRefineApparatus.png
electrorefining of copper process
Electrorefining
- Electrorefining of copper equation
- Electrorefining of copper ppt
- Electrorefining of copper Class 10
- Electrorefining of copper diagram
- Processing of copper electrorefining anode slime: a review - Taylor ...www.tandfonline.com › doi › pdf
- The copper anode contains many soluble and insoluble impurities. During the electrorefining insoluble anode impurities
- amounting about 3 to 5 kg/tonne of ...[PDF] Effect of Jet Flow between Electrodes on the Cathode Quality ... - MDPIwww.mdpi.com › pdf
- Oct 17
- 2018 · Copper electrorefining is the final process in copper pyrometallurgy to achieve the purity of the product. The copper ions dissolved from the blister ...[PDF] Primary Copper Smelter and Refinery as a Recycling Plant ... - MDPIwww.mdpi.com › pdf-vor
- Oct 24
- 2017 · The main process stages for copper production include feed preparation
- smelting
- converting
- fire-refining
- anode casting
- and electrorefining
- ...[PDF] FUNDAMENTAL STUDIES OF THE ANODIC BEHAVIOR OF ...open.library.ubc.ca › media › download › pdf
- The majority of electrolytic copper is produced by the copper electrorefining process in which impure copper is refined through dissolution from an impure copper ...Related searchesElectrorefining of copper anode mud
- Explain the process of electrorefining of copper
- Electrorefining of copper metal
- Electrorefining of gold
- Explain the process of electrorefining with an example
- Electrowinning of copper Pdf
- Why is impure copper used as an anode in electrorefining
- Electrolytic refining of copper
electrorefining of copper reaction
[PDF] Research Article The Effective Electrolytic Recovery of Dilute - Core
- Electrorefining of copper metal
- Electrorefining of lead
- Electrorefining of iron
- Copper electrowinning process
- [PDF] Understanding Impurities in copper electrometallurgy - Scholars' Minescholarsmine.mst.edu › cgi › viewcontent
- impurities in copper electrowinning and electrorefining. The first part ... over blast furnace or reverbatory furnace techniques by using the exothermic reaction.[PDF] Article (PDF) - journalssystem.comwww.journalssystem.com › ppmp › pdf-51298-2520 › filename=Cop...
- Active nitrate ion reduction reactions catalyzed by copper ions are suggested to ... In electrorefining of copper
- this requires new techniques and new means.Aqueous Processing of Metals - Wiley Online Libraryonlinelibrary.wiley.com › doi › 9783527610426.bard050401 › pdf
- 1 g of copper it is necessary to use 2 × ... reactions of interest in metal electrolysis [1]. Reaction. Standard electrode ... the electrorefining of copper the equilib-.[PDF] Research Article The Effective Electrolytic Recovery of Dilute ... - Corecore.ac.uk › download › pdf
- reduced in the electrical chemical reaction. The predominant cathode reactions during copper electrowinning from acid sulphate electrolytes can be represented ...Related searchesElectrowinning and electrorefining
- Why is impure copper used as an anode in electrorefining
- Electrowinning of copper ppt
- Electro refining method
- Electrowinning of nickel
- Principle of electrorefining
- Electrolytic copper
- Electrorefining furnace
électrotechnique exercices corrigés pdf
[PDF] CORRIGÉ DES EXERCICES DU CHAPITRE 2
- électrotechnique exercices corrigés pdf
- electrotechnique exercices corrigés pdf
- electrotechnique exercices corrigés
- electrotechnique exercices corrigés transformateur
- électrotechnique exercices corrigés
- materiaux electrotechnique exercices corrigés
- electrotechnique cours et exercices corrigés pdf
- circuit magnetique electrotechnique exercices corrigés
elementary art lesson plans pdf
[PDF] Lesson Plan
- Middle School Art lessons PDF
- Art projects PDF
- Teaching art to elementary students
- Art lesson plans for elementary
- Texture Art Lessons For Elementary - yb.tlwww.yb.tl › texture_art_lessons_for_elementary
- Jan 28
- 2008 · File Type PDF Texture Art Lessons For. Elementary. Texture Art Lessons For Elementary Art Texture. Lesson Plan for Elementary School ...[PDF] Lesson Plan Using Artistic Ways of Thinking Art methods ... - ERICfiles.eric.ed.gov › fulltext
- Using art making as the exemplar
- a lesson plan expanding the art knowledge base thereby allowing children to think like artists is ... school art lessons; therefore
- he suggested one of Sir ... portals/0/pdf/about/twb/TWB_Color_Chart. pdf. Payne ...[PDF] arts lesson plan template - Teacherplanet.comwww.teacherplanet.com › sites › default › files › lessons_pdfs › Simpl...
- Several varied-‐minute lessons within a unit adding up to two-‐hours? Illinois State Arts Standard: Select the appropriate standard for this lesson. □ 25 – ...[PDF] Lesson Planwww.birdvilleschools.net › lib › Centricity › Domain › elements_of_art
- Course Title: Principles of AAVTC. Session Title: Elements of Art. Lesson Duration: One class period. 1. Cover the elements of art: Line
- Shape
- Color
- Texture
- ...Related searchesLesson plan for art and craft class
- Free Art Worksheets PDF
- High School Art Lesson Plans PDF
- Elements and Principles of art Lesson Plans
- Visual arts lesson plans
- Grade 1 art lesson Plans
- Introduction to art and design PDF
- Art Worksheets for Middle School PDF